Section 3: Genomic Characterization
Section 1: Basic Characterization
Section 2: Pluripotency and the Undifferentiated State
Section 3: Genomic Characterization
Section 4: Stem Cell-based Model Systems
Appendix 1: Recommended Standard Characterization of Stem Cells
Appendix 2: Nomenclature Criteria
Appendix 3: Cell Culture Hygiene Practices
Appendix 5: Assessment of Methods for Genetic Analysis
Appendix 6: Reporting Practices for Publishing Results with Human Pluripotent and Tissue Stem
Stem cells are subject to the acquisition of genetic changes in culture (Draper et al., 2004; Mayshar et al., 2010; The International Stem Cell Initiative, 2011; Weissbein et al., 2014). Although variants initially appear as a single abnormal cell, their level of mosaicism can change over time, depending on the properties they confer upon cells. For example, variant cells that possess a selective growth advantage can rapidly expand in culture and outcompete wild-type cells to dominate cultures (Draper et al., 2004; Olariu et al., 2010; Price et al., 2021). Apart from a growth advantage, genetic changes can alter many different aspects of stem cell phenotype and behavior (Barbaric et al., 2014; Ben-David et al., 2014; Markouli et al., 2019). Moreover, culture-acquired genetic changes may also impact the behavior of differentiated cells derived from variant stem cells. By changing properties of cells, genetic alterations can affect reproducibility and repeatability of results obtained from stem cells and their differentiated derivatives. Hence, this section advocates monitoring of stocks and cultures of stem cells for the presence of culture-acquired genetic changes.
Assessing Genetic Status
Recommendation 3 .1 .1: Cultures should be monitored for the presence of culture- acquired genetic changes as these can have myriad irreversible effects on stem cells and their differentiated progeny, such as altered growth rate, tumorigenicity, differentiation potential, and functionality, that may significantly impact the reproducibility and reliability of data collected.
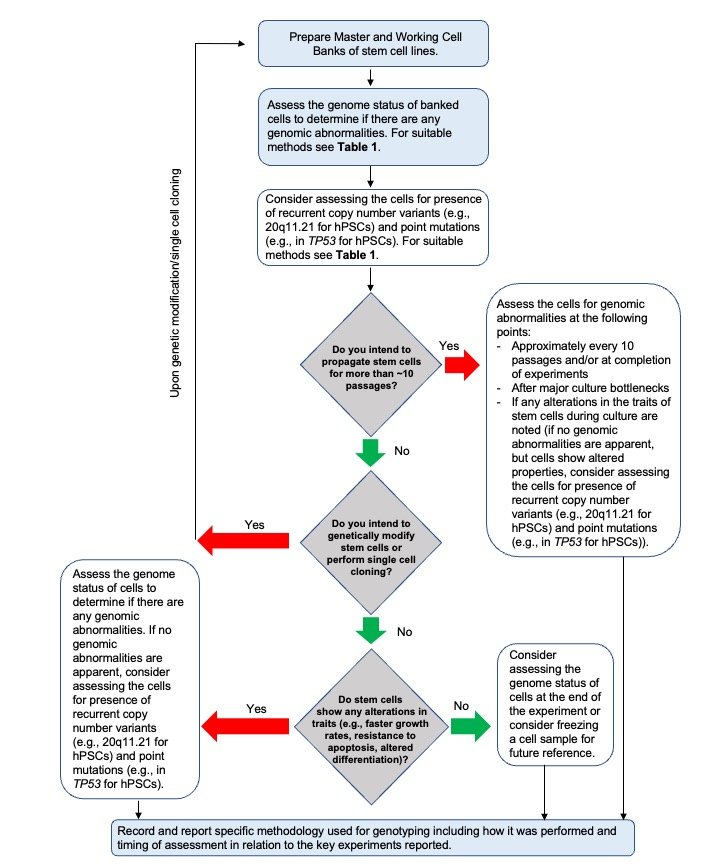
All cell cultures are vulnerable to cultured-acquired genetic changes and should be monitored to ensure the consistency of the genetic composition over time. Human PSCs in particular are prone to culture-acquired genetic changes at the chromosomal, sub-chromosomal and nucleotide levels (reviewed in Halliwell et al., 2020). Some of the commonly acquired genetic changes in hPSCs include, but are not limited to, gains of chromosomes or parts of chromosomes 1, 12, 17, 20 and X (Draper et al., 2004; Baker et al., 2016) and single nucleotide variants in TP53 (Merkle, et al., 2017; Lezmi and Benvenisty, 2021). Thus, routine monitoring of hPSCs should entail analysis of the chromosomal complement to detect numerical and structural aberrations. Since many recurrent culture- acquired changes in hPSCs known to date are numerical and structural abnormalities, this analysis is expected to be sufficient to detect most large aberrations likely to be encountered. However, researchers should risk- assess the need for higher resolution detection methods to identify any recurrent changes in hPSCs that are not detectable by analysis of the chromosomal complement, such as small copy number variants (e.g., 20q11.21 CNV) and single nucleotide variants, including those in TP53 and additional cancer-related genes. For example, additional monitoring may be warranted if any alterations are observed in the traits of hPSCs during culture, such as changes in growth patterns or differentiation ability. Moreover, specific applications (i.e., establishment of new culture conditions, reprogramming methods or preclinical work) may also require more extensive monitoring for the presence of genetic changes.
Although a range of methods for assessment of genetic changes is available, currently no single method detects all types of genetic changes with equally high sensitivity and specificity. Thus, the specificity and detection limits of the assays should be considered when choosing specific methods for assessment of cultures and interpreting negative results, i.e., variants may be present at a mosaicism or resolution below levels of detection for a given assay (Appendix 5, Table A5.1). Moreover, this area is constantly evolving and therefore the particular changes to focus on and the most appropriate methods to use should be subject to regular review. Some examples of presently relevant recurrent genetic changes in hPSCs are listed in Table A5.2 (Appendix 5) including some of their known phenotypic effects and suitable methods for detecting them.
Timing of Assessment
Recommendation 3 .2 .1: Master and Working cell banks should be evaluated to determine their genetic status.
A crucial timepoint for genetic assessment is during the preparation of the master and working cell banks. Cell lines are more likely to acquire genetic changes after a culture bottleneck, such as cloning events or gene editing. Ideally, master and working cell banks are created after such interventions, following genetic assessment. No method for detection of genetic changes can detect low levels of variant cells. Since the common recurrent variants provide the cells with a growth advantage, variants present below the level of detection in an initial screen may become detectable several passages later (e.g., see Olariu et al., 2010).
Recommendation 3 .2 .2: Monitoring for genetic changes should ideally cover the timespan of experiments.
Cells carrying variants providing a selective advantage can overtake a culture rapidly, often within 5-10 passages (Avery et al., 2013; Olariu et al., 2010). Therefore, not using cells that were drawn from the tested bank beyond passage 10 after thawing significantly decreases the risks of genetic drift. To avoid unnoticed genetic drift of the cell cultures used in experiments, there are several timepoints when genetic monitoring should ideally be carried out:
1) before starting the experiments (e.g., on the master or working cell banks).
2) during experiments (e.g., approximately every 10 passages*) to aid timely detection of genetic changes and/or on completion of experiments to assess whether the cell cultures used in experiments had retained their genetic integrity throughout the duration of the experiments.
3) after major culture bottlenecks, as these increase the chance of clonal expansion of genetically abnormal cells.
4) If any alterations are observed in the growth characteristics of stem cells during culture or in their patterns of differentiation, assessing the presence of genetic changes is recommended to check whether these alterations are caused by culture-acquired genetic changes. If no karyotypic abnormalities are apparent, but cells show altered properties, assessing the cells for presence of recurrent copy number variants not readily detected by karyotype analysis (e.g., 20q11.21 for hPSCs) and point mutations (e.g., in TP53 for hPSCs) is recommended.
*Passage is used here as it is a widely used and long established, convenient measure of population growth per passage, but the number of population doublings is affected by the split ratio and the cell cycle time, so it is advisable to consider these factors when choosing the interval between monitoring).
Recommendation 3 .2 .3: When a new cell line or derivative is generated by modifying culture conditions, implementing new reprogramming techniques, or after performing complex genome or epigenome manipulation, the cells should be evaluated for genetic changes after the intervention.
Analyzing cell material before and after generation of a new cell line or derivative enables comparison of genomic profiles and identification of newly introduced variants versus variants which were already present in the original cell material (Steeg et al., 2021).
For an overview of when and how to assess the genomic status of stem cells, see Figure 3 (below).
The ISSCR's Standards for Human Stem Cell Use in Research are strictly copyrighted by the society. No part of this document may be produced in any form without written permission of The International Society for Stem Cell Research. Contact isscr@isscr.org for more information.
©2023 by The International Society for Stem Cell Research. All rights reserved.